Lysosomes are critical organelles that perform a variety of cellular functions. They act as end stage degradative compartments, dealing with inputs from endocytic, phagocytic and autophagic routes, in order to maintain protein and organelle homeostasis and combat extracellular pathogens. Lysosomes also operate as signalling hubs, integrating cellular nutrient status and cell stress responses. Lysosome dysfunction is associated with many age-related declines in cellular function and the progression of organismal pathologies. The Florey lab is interested in exploring ways to harness novel mechanisms to boost lysosome function.
Canonical (macro)autophagy, is an intracellular degradation process that sequesters proteins or organelles in the cytosol and delivers them to the lysosome. This is a fundamental pathway, critical in maintaining cellular fitness and energy homeostasis. A hallmark feature of the pathway is a unique ubiquitin-like conjugation of ATG8 proteins to the lipid phosphatidylethanolamine in forming double-membrane autophagosomes. Recently, a distinct non-canonical autophagy pathway was identified that utilises some of the autophagy machinery to target the Conjugation of ATG8 to Single-Membranes (CASM).
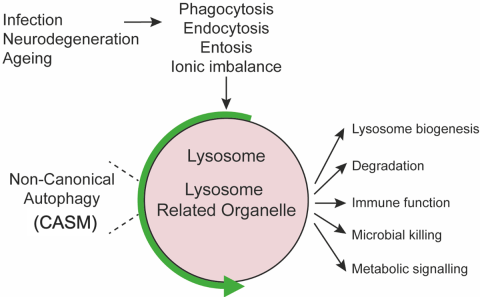
A diverse variety of stimuli, including influenza infection, apoptotic cell or pathogen phagocytosis, endocytosis and pharmacological disruption of vesicle pH, have been shown to induce CASM on endolysosomal vesicles. (Fig. 2) Non-canonical autophagy has been implicated in regulating important immune responses, such as the inflammatory signature generated in macrophage and dendritic cells upon infection or clearance of dead cells. Disruption of this can result in altered cytokine responses and auto-immune consequences.
The Florey lab has established novel techniques and reagents to dissect and interrogate the non-canonical autophagy pathway, and determine its functions and underlying mechanisms. This included the development of a new knock-in ATG16L1 point mutant mouse strain, which is selectively deficient for non-canonical autophagy.
We have a special interest in the role of CASM during phagocytosis and lysosome biology, and use state-of-the art fluorescent and electron microscopy and mass spectrometry coupled with a variety of different cellular systems, to gain insights into the regulation and function of CASM.